Working Party on Quality by Design - WP QbD
Welcome to the website of the EFCE Working Party on Quality by Design (WP QbD).
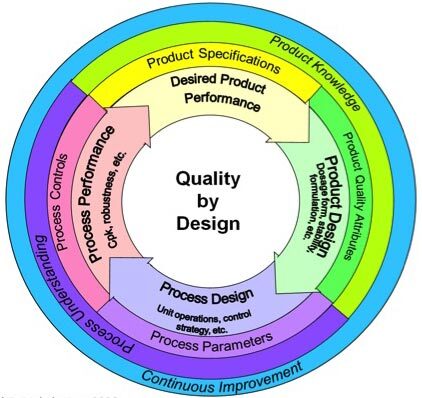
Quality by Design
- is "A systematic approach to development that begins with predefined objectives and emphasizes product and process understanding based on sound science and quality risk management"
- is a quality management method for pharmaceutical production industry
- is an evolving international standard for pharmaceutical development of APIs and products following: ICH-Guidelines Q8, Q9, Q10, Q11 and Q12; GAMP GMP Guidelines and Annexes; Guidelines towards systematic development process
- uses Quality Risk Management and scientific understanding for selection, specification and control of (1) critical Process Parameters (cPPs) & critical Material Attributes (cMAs) (2) affecting critical Quality Attributes(CQAs)
- provides guidelines for compiling data/information in relevant parts of the application file
- is applied to and affects life cycle management in order to Decrease Time-to-Market; Optimize quality and costs; Less OOS; Control strategy; Knowledge-management; Data Integrity; Regulatory Flexibility; Post Approval Change Management Protocol; Real Time release Testing
Current activities
Introduction
Biochemical processes producing value-added-products are and will remain critical to Europe’s competitiveness and, more importantly, essential to addressing important societal challenges, such as health and food. However, bioprocess development is often excessively long with a very high failure rate. Furthermore bioprocesses of industrial biotechnology producing chemical building blocks as well as technologies for the production of biosimilars also i) receive increased attention of the regulatory framework and ii) urge for more effectiveness in the process and product lifecycle, aiming at reducing cost of goods and optimizing productivity. In order to accelerate process development, to ensure scalability as well as transferability of processes between sites or process platforms, process understanding and knowledge gathered from other products, sites and processes must be readily utilised.
Concept of Quality by Design (QbD) has been introduced over 10 years ago in the (bio)pharmaceutical industries. QbD allows manufacturers to introduce process improvements without regulatory oversight, subject to proof process understanding. However, the full benefits of QbD are not exploited for many reasons: Most of the reasons fundamentally include gathering and transferring knowledge.
Reference: GMP Navigator
Business Meeting
tba
Spotlight Talk
Mission, Aims and Objectives
MISSION of the Working Party
This Working Party on QbD wants to propose clear workflows, which focus on knowledge transfer, will facilitate more effective and time-efficient QbD implementation in practice. This includes a mind-set change from considering QbD only as a regulatory threat or requirement to using it as an opportunity.
The Working Party on QbD aims at emphasizing the benefits of the structured approach and methods of QbD beyond regulatory requirements. Hence, the main aim of the Working Party on QbD is to elaborate on clear best practice workflows which allow knowledge transfer by facilitating of information- and knowledge-exchange between experts from academia and industry.
What do we aim to deliver?
This Working Party on QbD will catalyse increased cross-fertilization between the biochemical engineering and the QbD communities and provide improved training opportunities for early career researchers in this important subject with the final goal of ensuring effectiveness of the product life cycle of innovative bioprocesses. The basis of this proposal is the strong drive to strengthen the cross-fertilisation of chemical and biochemical engineering community to ensure more effective translation of the QbD principles from research to manufacturing practice.
The specific objectives of Working Party on QbD are:
- Establishment of conference series or sessions in well-established conferences with high visibility.
- Establishment of a European conference series dedicated to process knowledge and manufacturing knowledge.
- Pan-European transectoral training schools of early career researchers in tools required for rapid process development and data science.
- Producing a series of position papers to be published in scientific and industrial literature raising awareness of issues and proposed solutions.
Major objectives of the Working Party
- To further promote the use of chemical engineering methods and tools in pharmaceutical process development and operation, as well as in pharmaceutical product design
- To offer a suitable forum for dissemination of research results and for exchange of ideas
- To coordinate research activities at the European level by identifying areas and problems that are important for development of novel and improved pharmaceutical production processes
- To promote cross-disciplinary exchange of knowledge and ideas
- To actively seek collaboration with other EFCE working parties to practically realize the activities which are required to reach some of the above-mentioned objectives in practice.
- To make recent developments in the area available through an electronic newsletter that will be published every six months.
Who is a member/ delegate of the Working Party?
|
Prof. Dr. Massimiliano Barolo |
|
Mr. Jan Cornevin |
|
Prof. Krist V. Gernaey |
|
Prof. Jarka Glassey |
|
Prof. Christoph Herwig |
|
Prof. Dr.-Ing. Norbert Kockmann |
|
Ing. Francesca Marrani |
|
Prof. Julian Morris |
|
Prof. Dr. Zoran Novak |
|
Dr.-Ing Joachim Ritter |
|
Dr. ir. Gerrit Westhoff |
|
Prof.dr.ir. Edwin Zondervan |
Previous activities
EveNts
Spotlight Talks
4 December 2020
Quality by Design tools for the Optimization of (BioH)Pharma Processes System Modelling, Machine Learning and Digital Twins
Recording on YouTube
24 May 2023
Successful implementation of quality by design along the product life cycle – industrial use cases
Web-Seminar Series in Quality by Design (QbD)
Thursday, 24 September 2020, 16:00 CET
Title: Accelerating clone and strain selection using shake flasks, online monitoring and advanced data science - novel QbD approaches in early bioprocess development
Speaker: Barbara Pretzner, Werum IT Solutions, Vienna, Austria;Rüdiger Maschke, ZHAW, Wädenswil, Switzerland; Gernot John, Presens, Regensburg, Germany
Tuesday, 6 October 2020, 16:00 CET
Title: Accelerating vaccines process development and industrialization: can digitalization and in silico modeling help?
Speaker: Emanuele Tomba, GSK, Siena, Italy (ABSTRACT)
Thursday, 29 October 2020, 16:00 CET
Title: An integrated QbD solution for deploying Digital Twins and PAT for Control of Inclusion Body Refolding Processes
Speakers: Krisztina Koczka, Bilfinger Industrietechnik, Salzburg, Austria; Lukas Veiter, Chase, Linz, Austria
4th PhD course - Measurement, modelling and control in biochemical engineering
July 3‐5, 2017, TU Wien, Vienna, Austria
A unique hands‐on course to provide insight into measurement, monitoring, modelling and control (M3C) applied to biochemical processes. The course is supported by the M3C section of ESBES, and by the Quality‐by‐Design Working Party of the EFCE. Three days of teaching and computer exercises. Course material and computer codes are supplemented with an additional set of reading material.
Lectures by an international team of experts: Christoph Herwig (TU Wien, Austria); Jarka Glassey (Newcastle University, UK); Bernd Hitzmann (University Hohenheim, Germany); Krist V. Gernaey (Technical University of Denmark, Denmark)
Please find more information under http://esbes.org/Courses.html
Papers published
"QbD - status-quo and challenges to further advances in process knowledge"
September 2017 to "The Chemical Engineer"
Conference contributions
APACT, April 2018, Newcastle, UK
Contribution of one full QbD Session on process robustness; 26th April 2018 Newcastle, UK
Talk on Process robustness by Dr. Zomer
Keynote on QbD driven continous biomanufacturing, by Prof Herwig
DECHEMA Jahrestagung Sept 2018, Aachen, DE
Session on process robustness, organized by Prof. Herwig
2nd European Forum on New Technologies in Chemical Engineering, Digitalisation in Chemical Engineering, March 2019, Frankfurt, DE
Talk on holistic control strategies for the whole product life cycle by Prof. Herwig
ESCAPE 29, June 2019, Eindhoven, NL
Contribution of one full QbD Session on QbD advancements