Working Party on Thermodynamics and Transport Properties
The Thermodynamics and Transport Properties Working Party is part of the European Federation of Chemical Engineering.
It represents the community of academics and industrials, both researchers and practicing engineers who have an interest in physicochemical properties of materials.
The winner of 2026 EFCE "Michael L. Michelsen" award is prof. Ioannis ECONOMOU
In a very competitive and high-level competition, the 2026 EFCE Michael L. Michelsen Award was given to Professor Ioannis Economou in recognition of his internationally renowned research, which combines in-depth knowledge of molecular thermodynamics with a variety of applications across materials, energy, and environmental engineering (e.g., CO₂ capture, green solvents, MOFs, ionic liquids, pharmaceuticals, and shale gas technologies). The award also recognizes his great commitment to the EFCE Working Party on Thermodynamics and Transport Properties, as its founding member and
inaugural Chair, as well as his strong commitment to education and training in the field in Greece, Qatar, and internationally.
The award comprises a certificate and EFCE press communication, and a monetary award of EUR 2500 which is intended to cover travel costs and conference fees to attend the 34 th European Symposium on Applied Thermodynamics - ESAT 2026 in Lisbon, Portugal, on 10-13 May 2026 where the award was presented. Furthermore, prof. Economou delivered a keynote lecture with title: "Multi-scale simulation of complex chemical systems: Structure and physical properties for novel material and process design”
The Award is fully financially sponsored by AVEVA.
More info here: https://efce.info/MichaelMichelsenAward_TTP.html
All award winners are listed in Awards-tab.
the NEXT Spotlight Talks session Will be Autumn 2026
Previous Spotlight Talks organized by the WP:
Thermodynamics for Zero-to-Low Emission Technologies Mini Series: Part 1. Heat Pumps, 17.3.2026, in YouTube.
Role of aplied thermodynamics in a changing world (sustainability, energy transitiont, circularity), 14.11.2024, in YouTube.
How industry is using applied thermodynamics (abstract), 15.3.2024, in YouTube.
A view on the future of applied thermodynamics (abstract), 5.6.2023, in YouTube.
Machine learning and data management for the estimation of fluid properties and phase equilibria (abstract), 25.11.2022, in YouTube.
Electrolyte Thermodynamics challenges from industrial needs to academic research (abstract), 19.5.2021, in YouTube.
Thermodynamic tools for CO2 capture (abstract), 3.12.2020, in YouTube.
Excellence Award in Thermodynamics and Transport Properties 2027: The call will open in autumn 2026
The Award recognises a PhD thesis and associated papers published in the preceding two-year period, which demonstrate an outstanding contribution to research and/or practice in thermodynamics and/or transport properties.
The previous award was given to Dr. Carlos Albà I Carriga at the International Conference on Properties and Phase Equilibria for Product and Process Design – PPEPPD 2025 which was held in Munich, Germany, on 11-15 May 2025. Congratulations! The Award was generously sponsored by IFP Energies nouvelles (IFPEN).
All award winners are listed in Awards-tab.
7th Industrial Use of Thermodynamics (IUT) at ESAT 2026:
Thermodynamic Principles for Sustainable and Circular Systems: Key Dos and DONTs
More info here.
Check "Industrial Perspectives" for info about the previous IUTs.
Article summarizing the previous IUT discussion "The thermodynamic pharma challenge: A cross-cutting perspective" available at https://doi.org/10.1016/j.ejps.2025.107178 (open access).
The Working Party is sad to announce the passing of one of its most valued and supportive members, Dr. Karel Aim, who was an Emeritus Scientist at The Czech Academy of Sciences in Prague.
He was a highly respected colleague and a generous member of our community, a warm and kind person, a true gentleman.
We would like to send our condolences to his family and remember him through the words of our colleagues.
“We have lost an outstanding member of the thermodynamics scientific community. Karel was an exceptionally kind and warm person. He always had a kind word to share and a gentle smile. May he rest in peace.” Epaminondas Voutsas
“I remember Karl as an enthusiastic colleague and friend, very knowledgeable, humble and willing to serve the scientific community also through political engagements. We will miss him” Jean-Charles De Hemptinne
“I first met him at DTU in the early 1980s and we shared many good times together in many places around the world, especially in Europe and the US. I will always remember his warm smile and enthusiasm for life and research.” John O’Connell
“An excellent scientist and a great member of our European thermodynamics community. A very pleasant colleague to interact with, a real gentleman.” Y. Economou
“He was a highly respected colleague and a generous member of our community, and his contributions to thermodynamics and to ESAT will be long remembered.” Jean-Noel Jaubert
“He was such a great colleague, a true gentleman and will be deeply missed” Amparo Galindo.
A special In Memoriam session was also held during ESAT 2026. The tribute was presented by Prof. Martin Lisal, who highlighted Karel Aim’s collaborative spirit, generosity, and constant support to colleagues and the scientific community. The session was warmly received by attendees and reflected the deep appreciation and respect held for him by the community. During the ceremony, a commemorative plaque was presented and will be delivered to his family.
Special Session in Honor of Professor Maurizio Fermeglia at ESAT 2026
On Sunday, February 25th 2024, professor Maurizio Fermeglia, a former rector of the University of Trieste and a very active member of the WP passed away unexpectedly at 68 years of age. His death came as a shock to all the WP members. We express our sincere condolences to his family, friends and collaborators and we will celebrate his achievements and scientific legacy in our future events.
Special session will be organized at ESAT 2026.
Opinion paper: "A view on the future of applied thermodynamics" based on industrial survey
"A view on the future of applied thermodynamics" is now available as open access paper. This opinion paper discusses and gives expert opinion on various topics raised in "Industrial Requirements for Thermodynamic and Transport Properties: 2020" (also open access).
Graphical abstract on the right from Ind. Eng. Chem. Res. 2022, 61, 39, 14664-14680. The authors thank Urška Linda Beuermann for the design of the graphical abstract.
Short summary of the paper in EFCE Newsletter December/2022.
The Properties of Gases and Liquids - 6th edition
Authors of this new edition are: J. Richard Elliott, Vladimir Diky, Thomas Knotts IV, W. Vincent Wilding
Recruitment Forum
What is thermodynamics?
Why thermodynamics and transport properties matter in process and product design?
In many industrial applications thermodynamics and transport properties are in key role in steady-state and dynamic simulations for development of new technologies as well as troubleshooting and optimization of existing processes. Bad choices in selecting thermodynamic methods or failures in the parametrization of the methods can have for example following consequences:
- not meeting the business case (for example running at lower than design capacity),
- product properties not at specification,
- exceeding the emission limits and
- having safety issues.
In product design, understanding on thermodynamics and transport properties speeds up the development of the new products and optimization of their properties.
In the parametrization of the models and methods, correct and accurate experimental data is extremely important. When experimental data is not available, predictive methods can be often applied.
Our working party focus areas are experimental measurements and modelling (both correlative and predictive) in the field of thermodynamics and transport properties.
The text below is based on our discussion in the 30th business meeting.
Why to be member of our working party
- Network with other thermodynamic experts and to be part of the international professional society.
- Know about the progress of the expertise of our group members, industrial needs and scientific advancements.
- Share and get new ideas. Catch opportunities for research collaboration.
- Recruitment: getting new employees, new PhD's and postdocs.
- Raise our profile in EFCE and national organizations.
- Rewarding to see us achieving the goals.
If you wish to follow our discussions, please join our LinkedIn group.
Objectives
- Create European thermodynamic experts’ network including academia, industry, young and experienced. Networking also outside Europe.
- Organize conferences and events.
- Propose awards to young academics and professionals to promote their careers.
- Share information in conferences and publications. Provide guidelines and best practises.
- Point out needs and issues within our field. Share our views and priorities on future developments required in the field.
- Increase the visibility and understanding of importance of thermodynamics and transport properties in the society and other engineering communities. Publish position papers.
- Share views and advance the educational aspects of thermodynamics so that it is more relevant for practicing engineers.
- Taking away the fear of thermodynamics.
Task Group on Industrial Perspectives
The task group aims at promoting synergy between industrial and academic research. To that end, two surveys of industrial needs have been performed and published: survey published 2010 and survey published 2021 (open access).
Inspired by the industrial surveys, an opinion paper titled "A View on the Future of Applied Thermodynamics" was prepared and published as an open access paper. The picture below (from Ind. Eng. Chem. Res. 2022, 61, 39, 14664-14680) summarizes the main messages of the paper.
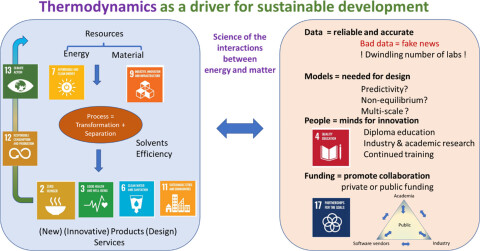
A continuous effort is required for communicating between academia and industry on the development of innovative approaches. This is why a new Symposium was launched, called Industrial Use of Thermodynamics (IUT). So far, following symposia were held:
- IUT1: InMoTher (Industrial use of Molecular Thermodynamics) was held in Lyon, France, in March 2012, with proceedings being published in OGST. On this occasion, a round table discussion was held focusing on the respective needs of industrial users, vendors and academics. Link to the open access publication.
- IUT2 was held in conjunction with the 27th European Symposium on Applied Thermodynamics (ESAT) in Eindhoven, July 2014. It was concluded with a round table discussion on needs for innovation as seen by industry. Link to the publication.
- IUT3 was held during the 10th European Congress of Chemical Engineering (ECCE10) in Nice, France, September 2015. It comprised 5 sessions, including a round table discussion on the use of thermodynamics for energy efficiency. Link to the publication.
- IUT 4 was a joined effort with the working party on Separation Processes, held during the 10th World Congress of Chemical Engineering in Barcelona, October 2017, and focused on data quality and assessment, validation methods and error propagation (open access publication).
- IUT5 was held in conjunction with the 31st European Symposium on Applied Thermodynamics in Paris, in July 2021. The focus was on electrolyte thermodynamics in industry. Link to the open access publication.
- IUT6 was held in conjuction with the ECCE 2023 in Berlin. The session was called: "The Pharma Challenge: a cross-cutting perspective from the WPs CAPE, Crystallization and Thermodynamics and Transport Properties" <Check out the leaflet>. Link to the open access publication.
- IUT7 was held in conjucntion with the ESAT 2026 in Lisbon. The session was called: Thermodynamic Principles for Sustainable and Circular Systems: Key Dos and DONTs. Opinion paper is in preparation.
Task Group on Education
Case Studies
In numerous industrial settings, the proper application of thermodynamics is an essential pre-requisite for good design and optimal operations. Not only is it necessary to better characterize the compounds and processes used in current industrial applications, but also it puts us in a better position, to develop more optimal processes and purpose-specific chemicals in order to address the global challenges of sustainability, climate change and energy usage.
Here, we have collected a number of real-life cases, that have occurred in industry, to illustrate not only the importance of correct thermodynamic description, but also the considerable subtlety needed, in the application of thermodynamic models, the choice of experimental data and understanding the results that come out of ubiquitous software packages.
- Heat Exchanger Calculations – example 1
- Heat Exchanger Calculations – example 2
- Air Humidity Calculations
- Simulator Data
- The use of UNIQUAC equation
- Dynamic viscosity of liquid mixture
Literature:
J. Gmehling, B. Kolbe, M. Kleiber, and J. Rarey, Chemical Thermodynamics: for Process Simulation, First Edition. © 2012 Wiley-VCH Verlag GmbH & Co. KGaA. Published 2012 by Wiley-VCH Verlag GmbH & Co. KGaA.
J. Gmehling, B. Kolbe, M. Kleiber, and J. Rarey, Chemical Thermodynamics: for Process Simulation, Second Edition. © 2019 Wiley-VCH Verlag GmbH & Co. KGaA. Published 2012 by Wiley-VCH Verlag GmbH & Co. KGaA.
J.C. de Hemptinne, J-M. Ledanois, P. Mougin, A. Barreau, How to select a thermodynamic model for process simulation: Problem analysis in three steps, Editions Technip, 2012 (ISBN 2710809494).
M. Kleiber, Process Engineering - Addressing the gap between study and chemical industry, De Gruyter, 2016.
Links
Useful web sites about thermodynamics
This incomplete list is a first result of the work of the members of the Working Party for Thermodynamics and Transport Properties within the EFCE who have collected a series of useful links, with emphasis on educational links. The list was then compiled by Peter Ahlström (University of Borås, Sweden). Among the contibutors are Prof Ralf Dohrn (Bayer Technology Services, Leverkusen, Germany) and Dr Juha-Pekka Pokki (Aalto University, Helsinki, Finland).
General sites
- There is a very good and comprehensive site by Prof Schwarzenhuber of the École des Mines for those of us who can read French http://nte.mines-albi.fr/ThermoPro/co/ThermoPro_html_1.html
- A long (somewhat personal) list of thermodynamics books is available at http://www.eoht.info/page/Thims’+thermodynamics+book+collection
- Marcello Castier’s YouThermo channel: https://www.youtube.com/channel/UCYNDgYUJKT6JcSzAoMitTEA
Textbook web sites
Normally these sites are accompanying the textbook and do not stand by themselves. Anyway a few of them follow here
- Elliot and Lira, Introductory Chemical Engineering Thermodynamics, 2nd ed. http://chethermo.net/
- The old site (for the 1st edition) http://www.egr.msu.edu/~lira/thermtxt.htm
- Sandler, S.I., Chemical and Engineering thermodynamics, 3rd ed, Wiley gives the supplementary Basic, Visual Basic or Mathcad files at the web page http://www.che.udel.edu/thermo/thermobook.htm
- Kontogeorgis handbook: http://bcs.wiley.com/he-bcs/Books?action=index&bcsId=5393&itemId=0470697261
- De Hemptinne website: https://view.genial.ly/5f2c0bef109c240d0308faa4/guide-applied-thermodynamics-ifp-school
- De Hemptinne's guide on thermodynamic model selection: https://view.genial.ly/617969334c81c40db6c41ec2/presentation-choose-your-model
Classes and lectures
- From MIT (lectures by W. Craig Carter) http://pruffle.mit.edu/3.00/Lecture_02_web/node2.html
Reference data
- Standard reference data of NIST (the program costs and at the the page there is a webshop) http://www.nist.gov/srd/
- There is however the web-book which contains a lot of free data http://webbook.nist.gov/ (requires a Java compatible web browser)
- The Dortmund databank online search (to see if the system is measured but the search does not provide the data itself). http://www.ddbst.com/en/online/Online_DDB_Form.php
- For those who want to program their code by themselves the UNIFAC, UNIFAC-DO, PSRK, etc. parameter matrixes are here http://www.ddbst.com/ChemThermo/Parameters/default.html
Open Source Thermodynamics Program Codes and Free software
- Marcello Castier at the Texas A&M University at Qatar has made several open source codes for thermodynamics to be used in MS Excel. They are described in the published paper
M. Castier, Mohamed M. Amer, XSEOS: An evolving tool for teaching chemical engineering thermodynamics, Education for Chemical Engineers, Volume 6, Issue 2, April 2011, Pages e62-e70, ISSN 1749-7728,
10.1016/j.ece.2010.12.002. http://www.sciencedirect.com/science/article/pii/S1749772811000030
Keywords: Equations of state; Excess properties; Phase equilibrium; Chemical equilibrium; Education - The original paper is XSEOS: An open software for chemical engineering thermodynamics Chem. Eng. Educ. 42 (2008), pp. 74-81, ISSN 0009-2479
The code should be possible to download from this link http://people.qatar.tamu.edu/marcelo.castier/index.htm
It contains several xls-files but the actual source code in Visual Basic is in XSEOS-Add-in.xla
This package was nice and pretty easy to follow but the link is presently defunct
Another one is from University of Valladolid in Spain and contains MATLAB files in *.m format.
Ángel Martín, María Dolores Bermejo, Fidel A. Mato, María José Cocero, Teaching advanced equations of state in applied thermodynamics courses using open source programs, Education for Chemical Engineers, Volume 6, Issue 4, December 2011, Pages e114-e121, ISSN 1749-7728, 10.1016/j.ece.2011.08.003.
http://www.sciencedirect.com/science/article/pii/S1749772811000091
Keywords: Phase equilibrium; Open source programs; Cubic equations of state; Statistical associating fluid theory SAFT; Group contribution; Education; MATLAB; Octave
It can be downloaded from the link given in the above *.pdf paper
http://iqtma.uva.es/hpp/en/download/download.html or
as a supplementary material of the paper. This is a zipped file that includes the *.m files. Those must be extracted from the *.rar file before seeing the *.m files. (using, e.g., 7-zip on a PC, for Ubuntu linux uncompression instructions arehere )
This package has a rather readable source code and a useful manual. - Clapeyron (formerly OpenSAFT) provides a framework for the development and use of fluid-thermodynamic models, including SAFT, cubic, activity, multi-parameter, and COSMO-SAC. Code available: https://github.com/ypaul21/Clapeyron.jl
- Thermodynamic databases supported by Reaktoro https://reaktoro.org/tutorials/basics/loading-databases.html
- NeqSim is a library for calculation of fluid behavior, phase equilibrium and process simulation. NeqSim can be used as a stand-alone tool via Excel or a web interface. It is integrated in computer programs via available interfaces in Java, Python, .NET and Matlab. The basis for NeqSim is fundamental mathematical models related to phase behaviour and physical properties of oil and gas. https://equinor.github.io/neqsimhome/
- Modeling Separation Processes (ChemSep): free ChemSep LITE with up to 40 components and 300 equilibrium stages using a database covering 400+ chemicals. http://chemsep.org/program/index.html. A free tool for performing binary VLE calculations and fitting model parameters is provided. Packages are CAPE-OPEN so that it can be used in any modern process simulator such as Pro/II, ASPEN, UNISIM Design, etc.
Working Party members
Working Party Board
SECRETARY:
Dr. Susanna Kuitunen
Neste, Finland
Contact

Members
AUSTRIA
CROATIA
CZECH REPUBLIC
DENMARK
ESTONIA
FINLAND
FRANCE
Dr. Marco Campestrini
Mines Paris, PSL University
Contact
(guest)

GERMANY
Dr. Emrah Altuntepe
Covestro
Contact
(delegate)

GREECE
HUNGARY
ISRAEL
ITALY
NETHERLANDS
NORWAY
PORTUGAL
Dr. José M.S. Fonseca
AVEVA
Contact
(guest)

ROMANIA
RUSSIA
SERBIA
SLOVAK REPUBLIC
SLOVENIA
SPAIN
SWEDEN
SWITZERLAND
UNITED KINGDOM
USA
Events
Upcoming Event
|
21/06/2026 - 24/06/2026 ECTP 2026Les Fontaines, France |
|
29/06/2026 - 30/06/2026 Sustainability Conference 2026Athens, Greece |
|
13/09/2026 - 15/09/2026 Thermodynamics 2026London, UK |
|
xx/05/2027 - xx/05/2027 ESAT 2027Marseille, France |
|
12/09/2027 - 16/09/2027 ECCE 2027Edinburgh, UK |
|
xx/xx/2027 - xx/xx/2027 EQUIFASE 2027x, Chile |
|
xx/xx/2028 - xx/xx/2028 PPEPPD 2028x, Chile |
|
26/08/2029 - 31/08/2029 WCCE 2029Prague, Czech Republic |
Past Events
|
13/05/2026 39th Working Party MeetingHybrid right after ESAT 2026 |
|
10/05/2026 - 13/05/2026 34th European Symposium of Applied Thermodynamics - ESAT 2026Lisbon, Portugal |
|
17/03/2026 Spotlight talks: Thermodynamics for Zero-to-Low Emission Technologies Mini Series: Part 1. Heat PumpsRecording on YouTube |
|
27/11/2025 38th Working Party MeetingOn-LINE |
|
08/09/2025 - 10/09/2025 ECCE & ECABLisbon, Portugal |
|
20/07/2025 - 24/07/2025 ICCT 2025Porto, Portugal |
|
14/07/2025 - 18/07/2025 WCCE12 & APCChE2025Beijing, China |
|
12/05/2025 37th Working Party MeetingHybrid, Bad Gögging, Germany (during PPEPPD 2025) |
|
11/05/2025 - 15/05/2025 Properties & Phase Equilibria for Product & Process Design - PPEPPD 2025Bad Gögging, Germany |
|
19/03/2025 - 21/03/2025 JEEP2025 : Journées d'Etude des Equilibres entre PhasesParis, France |
|
07/02/2025 36th Working Party MeetingON-LINE |
|
11/11/2024 - 12/11/2024 Annual meeting of process engineering and materials technologyDechema, Frankfurt am Main, Germany |
|
16/09/2024 - 18/09/2024 Sadi Carnot's Legacy - Celebrating the 200th anniversary of the 2nd law of thermodynamicsPalaiseau, France |
|
09/09/2024 - 12/09/2024 Equifase 2024Évora, Portugal |
04/09/2024 - 06/09/2024The 28th Thermodynamics Conference - Thermodynamics 2024Delft, Netherlands |
|
23/06/2024 - 28/06/2024 22nd Symposium on Thermophysical PropertiesBoulder, CO, USA |
|
13/06/2024 35th Working Party MeetingHYBRID meeting, Edinburgh, UK (right after ESAT) |
|
09/06/2024 - 12/06/2024 33rd European Symposium of Applied Thermodynamics - ESAT 2024Edinburgh, UK |
|
22/02/2024 34th Working Party MeetingON-LINE |
17/09/2023 - 21/09/2023
ECCE/ECAB 2023Berlin, Germany |
10/09/2023 - 13/09/2023
22nd European Conference on Thermophysical Properties - ECTP 2023Venice, Italy |
03/09/2023 - 06/09/2023
Heat Powered Cycles Conference (HPC)Edinburgh, uk |
22/05/2023
33rd Working Party MeetingHYBRID meeting, Tarragona, spain (during ppeppd 2023) |
21/05/2023 - 25/05/2023
Properties & Phase Equilibria for Product & Process Design - PPEPPD 2023Tarragona, Spain |
24/02/2023
32nd Working Party Meetinghybrid meeting, physical location IFPEN, Paris, France |
24/10/2022 - 27/10/2022XII Iberoamerican Conference on Phase Equilibria and Fluid Properties for Process Design (Equifase 2022) and XI Brazilian Conference on Applied Thermodynamics (CBTermo 2022)Campinas, Brazil |
07/09/2022 - 09/09/2022The 27th Thermodynamics Conference - Thermodynamics 2022 |
01/09/2022
31st Working Party Meeting, on-line |
22/08/2022 - 27/08/2022XXIII International Conference on Chemical Thermodynamics in Russia |
21/08/2022 - 25/08/202226th International Congress of Chemical and Process Engineering (CHISA) |
21/07/2022 - 22/07/2022The 11th Technical Thermodynamics: Thermophysical Properties and Energy Systems (THERMAM) |
07/07/2022 - 11/07/202232nd European Symposium of Applied Thermodynamics - ESAT 2022 |
25/03/2022
30th Working Party Meeting, Annual business meeting, on-line |
5/7/2021 - 9/7/2021
31st European Symposium of Applied Thermodynamics - ESAT 2021 video recordings |
Awards presented by the Working Party
Excellence Award in ThErmodynamics and Transport Properties
Lanched in 2009, the biennial Excellence Award recognises PhD theses or associated publication(s) which demonstrate the most outstanding contribution to research and/or practice in the field of thermodynamics and/or transport properties.
Previous Excellence Award winners are (more details below and here):
- Dr. Carlos Albà I Garriga (2025)
- Dr. Andrés Piña-Martinez (2023)
- Dr. Ailo Aasen (2021)
- Dr. Mónia Martins (2019)
- Prof. Dr. Øivind Wilhelmsen (2017)
- Dr. Bjørn Maribo-Mogensen (2015)
- Dr.-Ing. Christoph Held (2013)
- Dr. Maria Francisco Casal (2011)
- Dr. Eirini Karakatsani (2009)
The call for nominations for the 2025 Excellence Award in Thermodynamics and Transport Properties will open in autumn 2024.
“Michael L. Michelsen” Award
The “Michael L. Michelsen” Award (previously called "Distinguished Lecture on Thermodynamics and Transport Properties", renamed in Feb. 2021) aims to honour a senior member of the community that is active in a European institution. Launched in 2012, the award is presented every two years by the EFCE Working Party on Thermodynamics and Transport Properties.
The call for nominations for the 2024 Award is closed. It will be presented at ESAT 2024.
Previous lecturers/award winners are (more details below and here):
- Professor Ioannis Economou (2026)
- Professor Jean-Noël Jaubert (2024)
- Professor Signe Kjestrup (2022)
- Professor Gabriele Sadowski (2020)
- Professor Georgios Kontogeorgis (2018)
- Professor Cornelis J. Peters (2016)
- Professor Michael L. Michelsen (2014)
- Dr. Andreas Klamt (2012)
Dr. Carlos Albà I Garriga
Link to the press release (copied below).
Dr. Carlos Albà I Garriga is the winner of the 2025 EFCE Excellence Award in Thermodynamics and Transport Properties. He has been selected for his outstanding PhD thesis “Design of sustainable refrigerants by multiscale modelling”, performed at the University Rovira i Virgili under the supervision of Professor Fèlix L. Llovell Ferret.
Carlos Albà I Garriga obtained his MSc. in Chemical Engineering from the IQS School of Engineering in Barcelona, Spain, and his PhD in Nanoscience, Materials, and Chemical Engineering from the University Rovira i Virgili (URV), Tarragona, Spain. Since December 2024 he holds the position of R&D Engineer at GasN2 in Barcelona, Spain.
Carlos Albà I Garriga’s thesis addresses a critical environmental challenge of our time: the need for sustainable refrigerants to replace environmentally damaging compounds, the high-global-warming-potential (GWP) hydrofluorocarbons (HFCs). His work is rooted in advanced thermodynamic modeling, focusing on predicting and optimising properties critical to refrigeration cycles. By leveraging the polar soft-SAFT equation of state, Carlos has developed a robust multiscale modelling framework capable of accurately predicting a variety of thermodynamic properties across a wide range of conditions, reducing the dependence of costly experimental testing in many cases. He has extended the application of molecular theory to design 4th generation drop-in replacements to currently employed refrigerants. Using a multi-criteria assessment (4E analysis) on energy, exergy, environment and economy, he covered a wide range of aspects.
Dr. Albà’s research combines theoretical excellence with practical application, creating a multi-scale modelling framework that integrates molecular and process-level insights for the design and evaluation of new refrigerants. This work is timely, given the increasing global focus on reducing the environmental impact of cooling systems in the transportation and building sectors.
The jury of EFCE’s Thermodynamics and Transport Properties Working Party selected Dr. Albà I Garriga, among others, for the excellent technical quality of his work and the very clear presentation in his thesis.
His nominator, Professor Fèlix L. Llovell Ferret, said: “During his PhD, Carlos has conducted research stays at prestigious institutions, including ETH Zurich and Khalifa University, Abu Dhabi. These experiences have enriched his research by fostering international collaborations and exposing him to diverse methodologies and advanced facilities. His time at these world-renowned institutions has contributed to the development of innovative approaches in refrigerant design and enhanced the global impact of his research.”
The Excellence Award consists of a cash prize of €1500 plus the opportunity to attend the International Conference on Properties and Phase Equilibria for Product and Process Design – PPEPPD 2025 in Bad Gögging, Germany, on 11-15 May 2025, where the award will be presented.
The Excellence Award is generously sponsored by IFPEN and the EleTher research chair.

Dr. Andrés Piña-Martinez (2023)
Link to the press release (copied below).
His thesis, performed at Université de Lorraine, France, under the supervision of Professor Jean-Noël Jaubert, aimed at developing a methodology to determine the most appropriate working fluid for energy conversion machines with complex architecture that simultaneously produce cooling, heating and electric power. The use of such energy systems, which can be powered by renewable energy sources, should make it possible to achieve the objectives set by the European Union in terms of industry decarbonisation, and thus achieve carbon neutrality
by 2050.
The thesis included a comprehensive assessment of the accuracy of various equations of state (EoS) and different mixing rules for the calculation of phase equilibria and other properties of binary fluid mixtures. Dr. Piña-Martinez investigated several mixing rules that nicely integrate activity coefficient models (including Wilson, UNIQUAC and NRTL) within the cubic EoS. This work resulted in a large comprehensive set of cubic EoS with various mixing rules. Dr. Piña-Martinez developed all the necessary algorithms, codes and optimization techniques to perform the needed calculations. Based on his work, important conclusions were reached regarding the accuracy of the models and specific recommendations were developed for the use of these models for different classes of chemical mixtures. All the results obtained from the study of cubic and SAFT-type equations of state (EoS) could be used in the future in energy-related applications and in computer-aided process design.
The judging committee of EFCE’s Thermodynamics and Transport Properties Working Party selected Andrés Piña-Martinez for the Award based on the innovative approach to cubic equations of state, the extremely wide assessment of equation of state models and their modifications and the application of these models to energy technology applications, together with a high quality of the related publications.
Nominating him for the Award Jean-Noël Jaubert wrote: “… Andrés Piña-Martinez demonstrated he was a researcher brimming with ideas and tireless. The motivation he has shown and his outstanding abilities make him a scientist with a great future.” He also emphasized the excellent preparation and presentation of the doctoral thesis. The Excellence Award consists of a cash prize of €1500 plus the opportunity to attend the International Conference on Properties and Phase Equilibria for Product and Process Design – PPEPPD 2023 in Tarragona, Spain, on 21-25 May 2023, where the award will be presented. The Award lecture is scheduled for 23 May 2023.
The Excellence Award is generously sponsored by Bayer AG.
Currently (July, 2024), Andrés is assistant professor at Université de Lorraine, France. He is working on process systems engineering and more specifically on the development of computer-aided process synthesis approaches.

Dr. Ailo Aasen (2021)
His PhD thesis on "Bulk and interfacial thermodynamics of mixtures: From aqueous systems to ultracryogenic fluids", completed at the Department of Energy and Process Engineering, Faculty of Engineering, Norwegian University of Science and Technology (NTNU), Norway, under the supervision of Professor Øivind Wilhelmsen, achieved the best evaluation results in terms of dissemination of knowledge including quality of the publications and presentations, duration of the thesis, originality of the topic studied and of the methodology followed, innovation and industrial relevance, and scientific impact of the work.
Ailo Aasen obtained his MSc in Applied Physics and Mathematics from the Department of Mathematical Sciences, Faculty of Information Technology and Electrical Engineering, NTNU, Norway. Currently, he holds the position of research scientist and software developer for computational thermodynamics and fluid mechanics at SINTEF Energy Research.
His projects include: Thermodynamic modelling and equation-of-state development for fluid mixtures, with application to carbon capture and storage (CCS), and hydrogen liquefaction processes; Fluid-mechanical modelling for multiphase, multicomponent flow through nozzles, with special emphasis on choked flow; Unit modelling of ejectors for sizing and optimization; Designing tanks for liquid hydrogen; and Interpolation algorithms for computational thermodynamics
The award will be presented at the 31st European Symposium on Applied Thermodynamics - ESAT 2021 which take place online, from 4-7 July 2021.
Currently (August, 2024) Ailo is a research scientist at SINTEF Energy Research in Trondheim, Norway. His research focuses on the thermodynamics and process simulation of carbon capture and storage, as well as the technical and safety aspects of using liquid hydrogen and liquid ammonia as prospective energy carriers.

Dr. Mónia Martins (2019)
Dr. Mónia Martins has been awarded the 2019 EFCE Excellence Award in Thermodynamics and Transport Properties for her excellent PhD thesis “Studies for the Development of New Separation Processes with Terpenes and their Environmental Distribution”. Read the full Press release.

Prof. Dr. Øivind Wilhelmsen (2017)
PROF. DR. ØIVIND WILHELMSEN is the winner of the EFCE Excellence Award in Thermodynamics and Transport Properties 2017.
His PhD thesis on “Equilibrium and Non-equilibrium thermodynamics of planar and curved interfaces”, completed at the Norwegian University of Science and Technology (NTNU) supervision of Prof. Signe Kjelstrup, achieved the best evaluation results in terms of breadth and depth of the thesis, dissemination of knowledge, originality of the topic studied and of the methodology followed, innovation and industrial relevance, and scientific impact of the work.

Dr. Bjørn Maribo-Mogensen (2015)
Dr Maribo-Mogensen, now a Physical Property Specialist at Linde Engineering, Munich, Germany, successfully modelled an equation of state for electrolytes with applications in the oil and gas industry as part of his research.
He also developed a deeper understanding of the different models currently used for electrostatic interactions, comparing the Debye–Hückel model with the mean spherical approximation (MSA) theory.
Dr Maribo-Mogensen’s thesis has led to the development of engineering software that has substantial potential for industrial applications, such as describing the effects of electrolytes on natural gas sweetening, hydrates modelling and biofuels processing.
Dr Maribo-Mogensen said: “During the course of my PhD, I have been trying to find the right balance between making my research scientifically relevant and yet useful for industrial applications. Little did I know that the journey I started back in 2010 would lead to being the recipient of this Excellence Award."
“I am honoured to receive this recognition for my work which would not have been possible without the lasting support from my family, colleagues and supervisors. The award will motivate me to continue pursuing challenging goals and contribute to research in thermodynamics and transport properties.”
Professor Jean-Noel Jaubert, chair of the selection committee for EFCE’s Working Party on Thermodynamics and Transport Properties, said: “Dr Maribo-Mogensen’s PhD work is characterised by a pioneering, novel and highly independent work on electrolyte thermodynamics. The overall target has been to develop an electrolyte equation of state for application in the petroleum and chemical industries, including gas solubilities in mixed solvents containing salt and gas hydrate formation.
Currently (August 2024) Bjørn is Chief Technology Officer, Hafnium Labs ApS.

Dr.-Ing. Christoph Held (2013)
A four-year PhD research project has beaten seven other nominations for the prestigious Excellence Award awarded every two years by the European Federation of Chemical Engineering (EFCE) Working Party on Thermodynamics and Transport Properties.
Dr-Ing Christoph Held, 32, from the Technische Universität Dortmund, Germany, has been recognised for his thesis "Measuring and Modelling Thermodynamics Properties of Biological Solutions", which, on submission, received the highest possible grade of summa cum laude and has subsequently been published in nine peer-reviewed international journals.
Currently (August 2024) Christoph is an independent researcher and lecturer at the Lab of Thermodynamics at TU Dortmund University. He investigates phase equilibria, reaction equilibria and kinetics of electrolyte solutions and respective applications in technical machines (rotating packed beds). More details available here: https://th.bci.tu-dortmund.de/team/group-leaders/dr-ing-christoph-held/.

Dr. Maria Francisco Casal (2011)
Dr. Maria Francisco Casal, Spain, is the winner of the Excellence Award in Thermodynamics and Transport Properties 2011 of the European Federation of Chemical Engineering (EFCE). Her PhD thesis on “Desulfurization of fuel oils by solvent extraction using ionic liquids”,” completed at the University of Santiago de Compostela, Spain, under the supervision of professors Alberto Arce and Ana Soto, received the best evaluation in terms of dissemination of knowledge, originality of the topic, depth of the study, industrial relevance, and impact of the work.

Dr. Eirini Karakatsani (2009)
Dr. Eirini Karakatsani, Greece, is the prize winner of the Excellence Award in Thermodynamics and Transport Properties 2009 of the European Federation of Chemical Engineering (EFCE).
Her excellent PhD thesis on “Development and Evaluation of a New Equation of State for Polar Fluids: Pure Components and Mixtures”, completed at the National Technical University of Athens (NTUA) under the supervision of Dr. Ioannis G. Economou, Research Director at the National Center for Scientific Research (NCSR) “Demokritos”, received the best evaluation results in terms of dissemination of knowledge, originality of the topic, depth of the study, industrial relevance, and impact of the work.
Currently (August 2024) Eirini is principal scientist in Topsoe, Denmark. She is responsible for the existing proprietary thermodynamic programs of the company, as well as for new developments. She work individually, as part of a helpdesk team, but also as part of dedicated project teams.

Professor Ioannis Economou (2026)
In a very competitive and high-level competition, the 2026 EFCE Michael L. Michelsen Award was given to Professor Ioannis Economou in recognition of his internationally renowned research, which combines in-depth knowledge of molecular thermodynamics with a variety of applications across
materials, energy, and environmental engineering (e.g., CO₂ capture, green solvents, MOFs, ionic
liquids, pharmaceuticals, and shale gas technologies). The award also recognizes his great commitment to the EFCE Working Party on Thermodynamics and Transport Properties, as its founding member and inaugural Chair, as well as his strong commitment to education and training in the field in Greece, Qatar, and internationally. Read more.
The 2026 Michael L. Michelsen Awardee is generously sponsored by AVEVA.

Professor Jean-Noël Jaubert (2024)
Jean-Noël Jaubert has been named as the recipient of the prestigious Michael L. Michelsen Award for thermodynamics by the European Federation of Chemical Engineering (EFCE). Prof. Jaubert is a pioneer of applied thermodynamics and professor of chemical engineering and thermodynamics at the École Nationale Supérieure des Industries Chimiques (ENSIC), an elite school of chemical engineering at the University of Lorraine. The Michael L. Michelsen Award honours a senior member of the thermodynamics community and is presented by the EFCE Working Party on Thermodynamics and Transport Properties. Read more.

Professor Signe Kjelstrup (2022)
Professor Techn. Signe Kjelstrup, Norway, has been named the laureate of the 2022 EFCE Michael L. Michelsen Award. She was conferred the award by the EFCE Working Party on Thermodynamics and Transport Properties in recognition of her internationally renowned research in the area of non-equilibrium thermodynamics, with emphasis on entropy production minimization, electrochemical cell modelling, heterogeneous systems and nano-thermodynamics. Read more.

Professor Gabriele Sadowski (2020)
The 2020 Distinguished Lecture in Thermodynamics and Transport Properties has been awarded to Professor Gabriele Sadowski, TU Dortmund, Germany. The Federation’s Working Party on Thermodynamic and Transport Properties selected Gabriele Sadowski for her internationally recognised work related to advances of the statistical associating fluid theory (SAFT). The perturbed chain SAFT (PC-SAFT) approach, published in 2001 with Prof Joachim Groß, has had a vast impact in the capability to model complex fluids and is widely used all over the world, nowadays even in pharmaceutical industry. Read more.

Professor Georgios Kontogeorgis (2018)
“I am honoured for this recognition from the European Federation of Chemical Engineering / Working Party on Thermodynamics and Transport Properties. I see it as much more than a personal recognition but especially, as mentioned by EFCE itself, as a recognition of our efforts and leadership of industrial consortia which have thermodynamics as foundation”, says Georgios Kontogeorgis.
“With the recognition of the great results of Professor Georgios Kontogeorgis, DTU Chemical Engineering has consolidated our leading position in the important field of applied thermodynamics”, says Head of Department Kim Dam-Johansen.
The lecture was delivered at the 30th ESAT 2018 – European Symposium on Applied Thermodynamics, Prague, Czech Republic, June 10-13 2018.
Abstract:
Mysteries of Water Thermodynamics – Questions and some Answers
Water is the most important substance in the world; it covers two thirds of the Earth and our own cells include two thirds water by volume. Hundreds of books have been written about water and at the same time we know so little about it. In the words of Philip Ball, for many years consultant of Nature: “No one really understands water. It’s still a mystery” [1]. Water has more than 50 exceptional physico-chemical (thermodynamic) properties which are considered “anomalous” in the sense that no other liquids behave that way (trends with temperature, pressure and composition). Among the most exciting ones are the maximum of density at 4 oC, high values of heat capacity (stabilizing Earth’s climate) and surface tension (small insects walk on water) and maxima/minima of many thermodynamic properties as function of temperature e.g. the minimum hydrocarbon solubility in water at room temperature (related to the hydrophobic effect) and the speed of sound.
What is the true reason for all these ? Maybe hydrogen bonding “in some form” and especially the hydrogen bonding structure and its changes are the prevailing explanations. But which hydrogen bonding structure ?
Numerous theories for water structure have been presented but they are all up for debate. It is unclear whether liquid water maintains the tetrahedral structure (as we know it from ice) or whether it should best be described by a two-state model, where most molecules are in the form of rings or chains [3] and literature is full with heated discussions [1-2]. Effects of salts [4], surfaces and biomolecules on water structure are not well understood. Neither molecular simulation nor advanced experimental methods have provided full answers. Direct spectroscopic and other measurements could provide quantitative information on the degree of hydrogen bonding of water but they are not in good agreement with each other and these data can be interpreted in different ways [5,6]. Promising novel association theories have problems if an unclear picture of water is the input and it is not surprising that they cannot explain many of the anomalous properties of pure water and aqueous solutions. Moreover, novel theories giving new insight have been presented, most recently the “exclusion zone” concept pioneered by G. Pollack [7]. Where is the truth ? The purpose of this presentation is to illustrate some of the recent theories about water structure, their role in thermodynamics, try to answer some of the aforementioned questions and point out areas where further investigations are needed.
References
Ball, N., 2008. Water – an enduring mystery. Nature, 452, 291.
Ball, N., 2003. How to keep dry in water. Nature, 423, 25.
Wernet, Ph. et al., 2004. Science, 304, 995.
Frosch, M., Bilde, M., Nielsen, O.F., 2010. From Water Clustering to Osmotic Coefficients. J. Phys. Chem. A., 114, 11933.
Kontogeorgis, G.M., Tsivintzelis, I., von Solms N., Grenner, A., Bogh, D, Frost, M., Knage-Rasmussen, A., Economou, I.G., 2010. Use of monomer fraction data in the parametrization of association theories. Fluid Phase Equilibria, 296(2): 219-229.
Liang, X.D., Maribo-Mogensen, B., Tsivintzelis, I., Kontogeorgis, G.M., 2016. A comment on water’s structure using monomer fraction data and theories. Fluid Phase Equilibria, 407: 2-6
Pollack, G.H., 2013. The fourth phase of water. Beyond Solid, Liquid, and Vapor. Ebner & Sons Publishers, Seattle WA, USA.

Professor Cornelis J. Peters (2016)
Professor Peters has spent most of his career at Delft University of Technology, and continues now, after a rich contribution in The Netherlands, to mentor researchers and teachers in several universities worldwide (Petroleum Institute in Abu Dhabi, U. of Maryland, Colorado School of Mines, Petronas Technical University, Malaysia). He has been a pioneer in the design, development and optimization of new experimental methods to measure accurate thermodynamic and phase equilibrium properties. Reviews on high-pressure phase equilibria revealed that he has been one of the most active author worldwide in the last 30 years, producing highly valuable data and insight in phase behavior. In parallel, he has been a world leader in the application of theoretical models for complex fluids and molecular simulation methods to explain physical phenomena at different length and time scales.
His research has impact both in fundamental physical chemistry and thermodynamics but also in applications to industry. His most recent contributions refer to energy storage and production with emphasis on hydrogen technology, carbon capture and sequestration, novel technologies for seawater desalination using hydrates, design and optimization of new ionic liquids and deep eutectic solvents for gas separations and many others.
He has co-authored more than 250 peer-reviewed papers and 18 book chapters, he has authored / edited 6 books and has given more than 200 presentations (of which more than 50 were keynote / plenary talks) in international conferences, Universities and industry worldwide. His work has received more than 7,000 citations and has an H-index of 43. He held Professor / Visiting Professor positions in The Netherlands, Japan, Malaysia, Spain, UAE, UK and USA.
Professor Michael L. Michelsen (2014)
Professor Michael L. Michelsen, of the Technical University of Denmark (DTU), has been selected to deliver the latest European Federation of Chemical Engineering (EFCE) Distinguished Lecture.
The Federation’s Working Party on Thermodynamic and Transport Properties nominated Professor Michelsen in recognition of his outstanding work in the field of thermodynamics.
In a career spanning 40 years, Michelsen’s work has been influential in the energy and chemical industries, especially oil and gas exploration.
The award comprises of a certificate and €1,500 cash prize.

Dr. Andreas Klamt (2012)
The European Federation of Chemical Engineering (EFCE) announces that Dr Andreas Klamt has been selected to deliver the first Distinguished Lecture in Thermodynamics and Transport Properties.
Dr Klamt’s nomination comes in recognition of his pioneering and influencing work in applied chemical engineering thermodynamics via the development of the COSMO and COSMO-RS methods.
Based on his work at the Max-Planck-Institute for Metal Research in Stuttgart, he received his PhD in 1987 with a thesis on States of small positively charged particles in metals. His career subsequently took him to Bayer AG in Leverkusen where he worked on Computational Chemistry projects, eventually specializing in solvation and physical property calculation. He developed the methods COSMO and COSMO-RS, now widely used in the computational chemistry and chemical & biochemical engineering communities.
Currently a lecturer at the Institute of Physical and Theoretical Chemistry at the University of Regensburg, Dr Klamt teaches block courses on solvation modelling and physical property predictions in solution. In 2012 he was appointed as honorary professor.
Dr Klamt will deliver the Distinguished Lecture at the 26th European Symposium on Applied Thermodynamics, Potsdam, Germany, on 7 October 2012.
Launched by the EFCE Working Party on Thermodynamics and Transport Properties, this lecture will in future be a biennial presentation that recognizes outstanding research and achievements in this specialist field.
Members area
The following pages are restricted to members of the EFCE Working Party (EFCE Section).
EFCE is working with the DECHEMA Community-Tool. If you attended former events of the EFCE (e.g. WCCE10, Loss Prevention 2019) or DECHEMA, your address may be registered already. Please fill in these login details below.
Recruitment Forum
Please advertise open position at our LinkedIn group.


















